Vsepr shapes of bent1/14/2024  They have central angles from 104° to 109.5°, where the latter is consistent with a simplistic theory which predicts the tetrahedral symmetry of four sp 3 hybridised orbitals. There are several variants of bending, where the most common is AX 2E 2 where two covalent bonds and two lone pairs of the central atom (A) form a complete 8-electron shell. This geometry is almost always consistent with VSEPR theory, which usually explains non- collinearity of atoms with a presence of lone pairs. Nonlinear geometry is commonly observed for other triatomic molecules and ions containing only main group elements, prominent examples being nitrogen dioxide (NO 2), sulfur dichloride (SCl 2), and methylene (CH 2). The bond angle between the two hydrogen atoms is approximately 104.45°. Water (H 2O) is an example of a bent molecule, as well as its analogues. Certain atoms, such as oxygen, will almost always set their two (or more) covalent bonds in non-collinear directions due to their electron configuration. 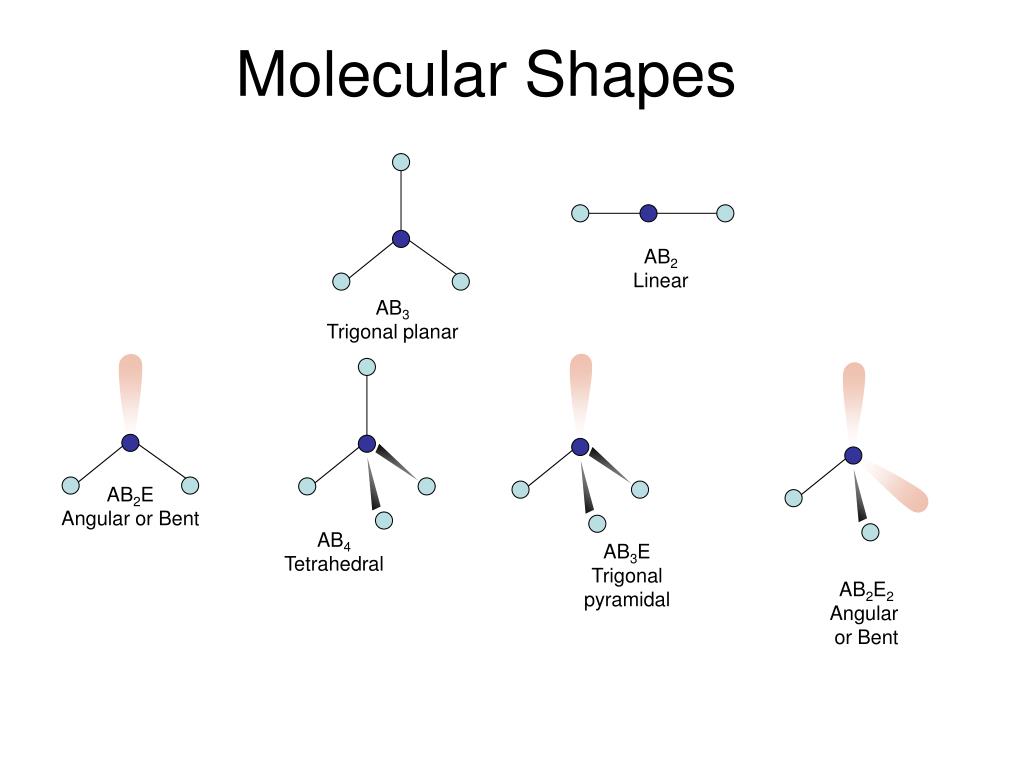
In chemistry, molecules with a non-collinear arrangement of two adjacent bonds have bent molecular geometry, also known as angular or V-shaped. See the site’s content usage guidelines.Oxygen difluoride, an example of a molecule with the bent coordination geometry. The graphic in this article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. If you want to learn more about working out the shapes of molecules using VSEPR, check out the provided links in the ‘further reading’ section below. This leads to the range of different shapes for the same number of electron pairs, as indicated in the graphic. Consequently, if they are present in a molecule, they can have an effect on the shape, taking up a space that would usually be occupied by a bonding pair, and making the angles between the other bonds in the molecule marginally smaller (by approximately 2.5˚ per lone pair present around the central atom). The presence of electrons that are not involved in bonding around the central atom, so-called ‘lone pairs’, are closer to the central atom, and as such repel electron pairs around them more strongly. Note that this method assumes that only single bonds are present in the molecule these are formed using a pair of shared electrons (1 from each atom), whereas double bonds involve two pairs of shared electrons.Ī complication of VSEPR is that all electrons do not repel equally. 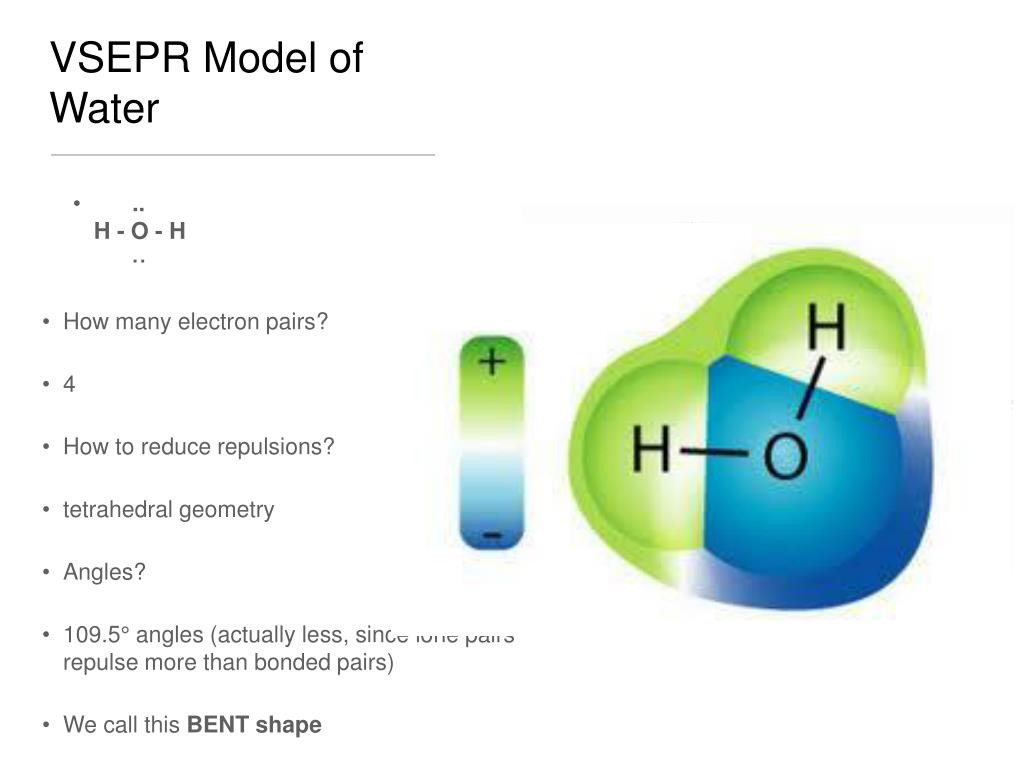
This can be determined in a few short steps, using the method provided in the graphic. In order to work out the shape adopted, we simply need to find the number of electron pairs surrounding the central atom in a molecule. However, it generally gives good predictions for compounds of main group elements. It doesn’t take into account factors such as the size of bonded atoms or groups, and as such doesn’t always predict the shape of certain compounds correctly, in particular those of transition metals. 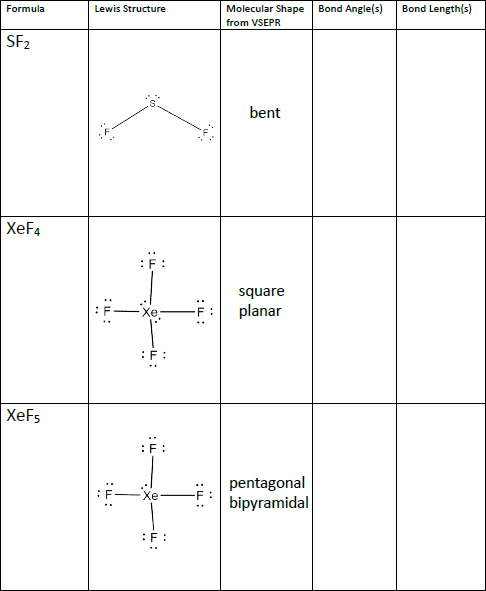
VSEPR works on the assumption that the shape adopted is that which minimises repulsions between the electron pairs in the molecule. One for the chemistry students (and teachers!) out there today, with a look at how we can work out the shapes of some simple molecules using Valence Shell Electron Pair Repulsion (VSEPR) theory. These shapes are decided by the arrangement of electrons around the central atom in the molecule. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
